
Based on their structures, explain why sodium oxide, silicon dioxide and carbon dioxide have different melting points. Here are 6 real student answers. - ppt download
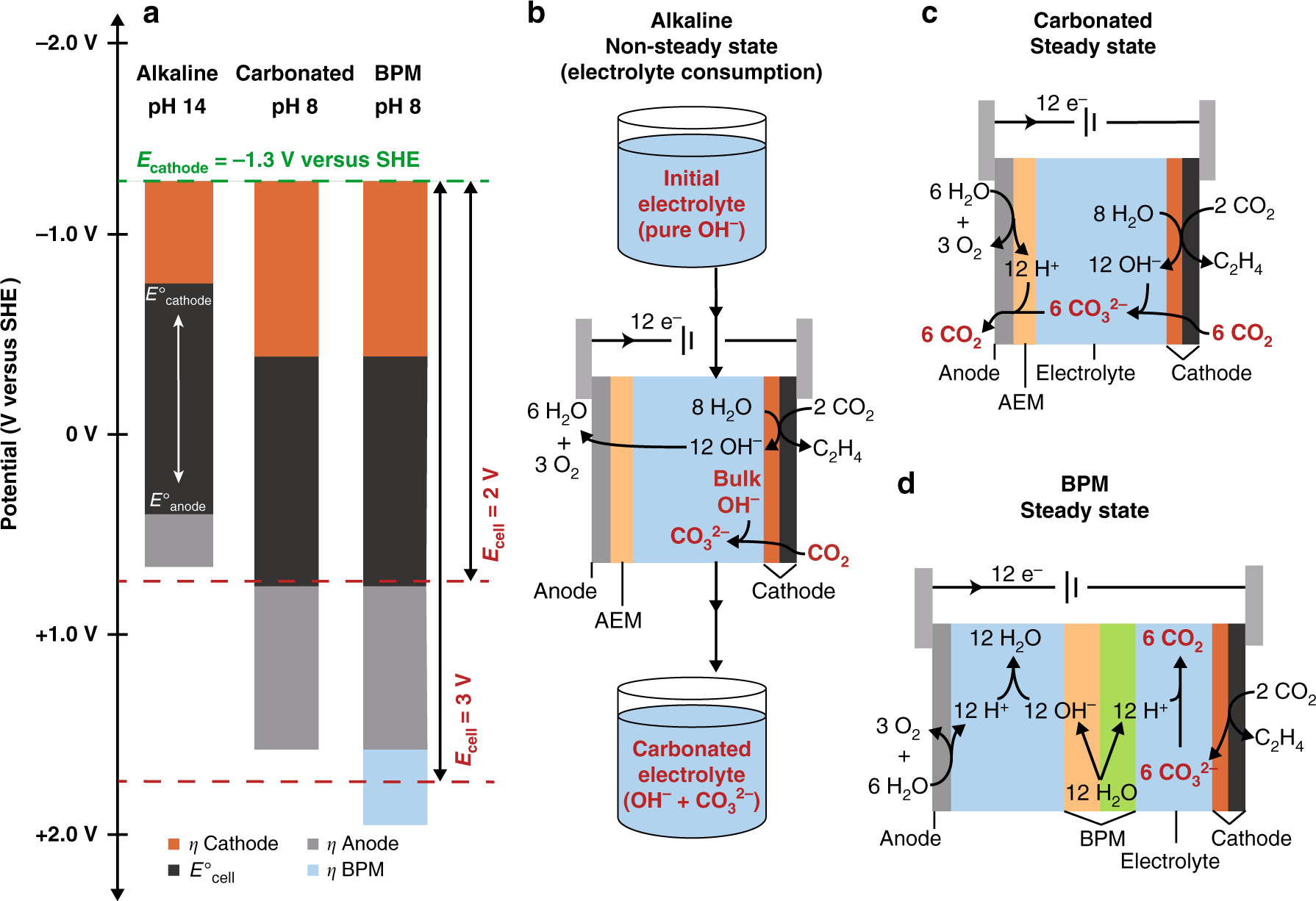
The future of low-temperature carbon dioxide electrolysis depends on solving one basic problem | Nature Communications

High-pressure melting equilibrium of chiral compounds: A practical study on chlorinated mandelic acids under carbon dioxide atmosphere - ScienceDirect
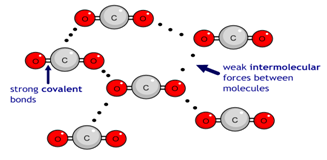
1:47 explain why substances with a simple molecular structures are gases or liquids, or solids with low melting and boiling points. The term intermolecular forces of attraction can be used to represent

Based on their structures, explain why sodium oxide, silicon dioxide and carbon dioxide have different melting points. Here are 6 real student answers. - ppt download

Do Now Check your class book for comments and green stickers. Sign and date these stickers and carry out any actions points that have been written for. - ppt download

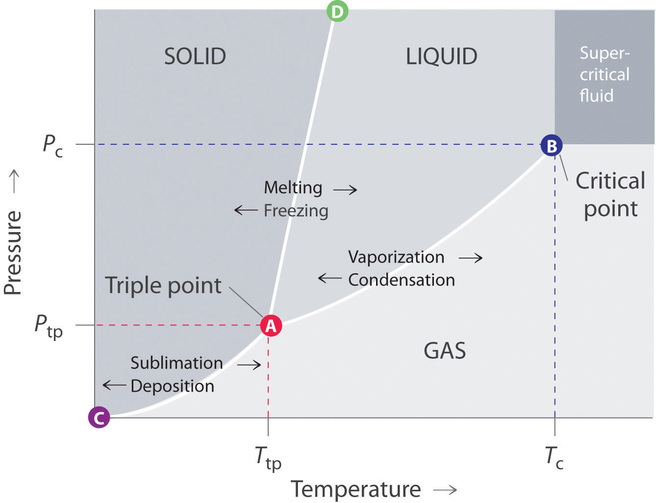



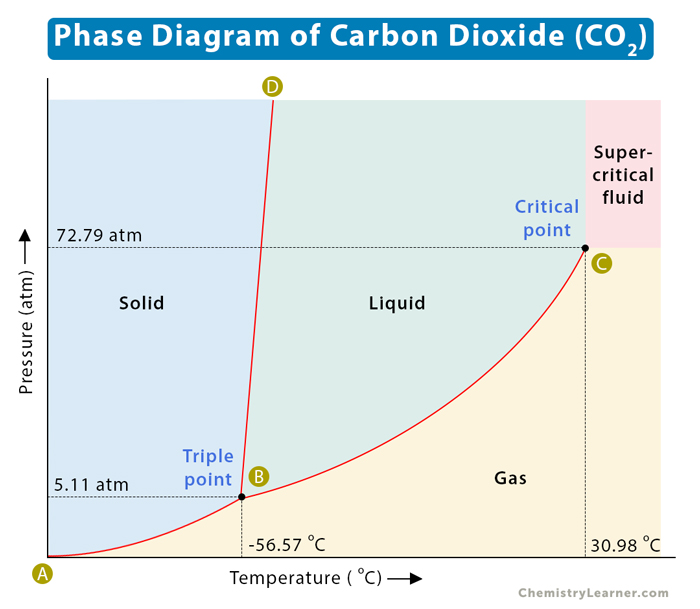
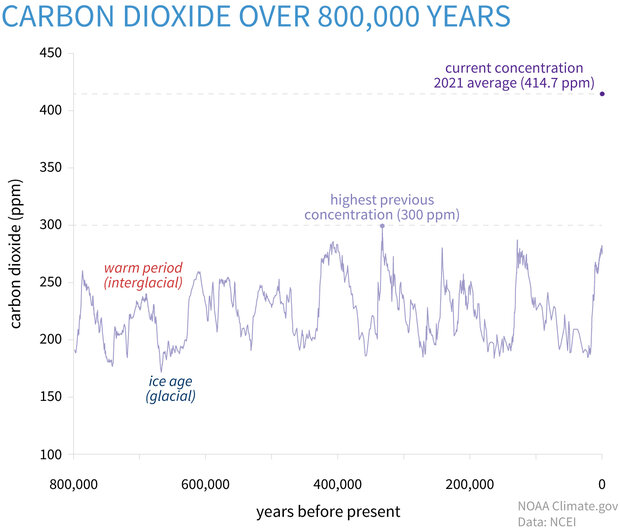
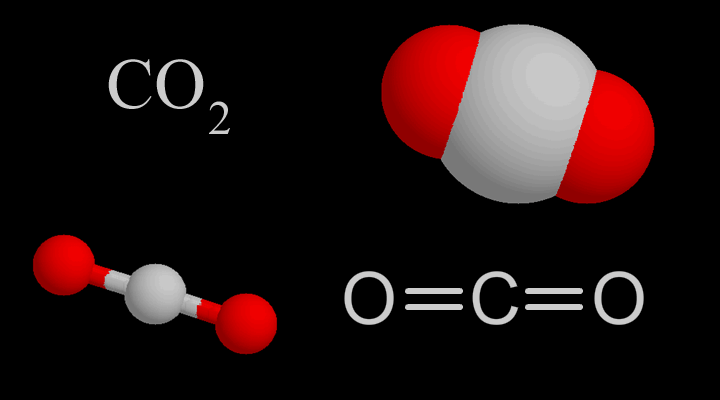

![Boiling temperature vs. pressure for CO2 [6] | Download Scientific Diagram Boiling temperature vs. pressure for CO2 [6] | Download Scientific Diagram](https://www.researchgate.net/publication/266441851/figure/fig4/AS:456125523992580@1485760207188/Boiling-temperature-vs-pressure-for-CO2-6.png)


