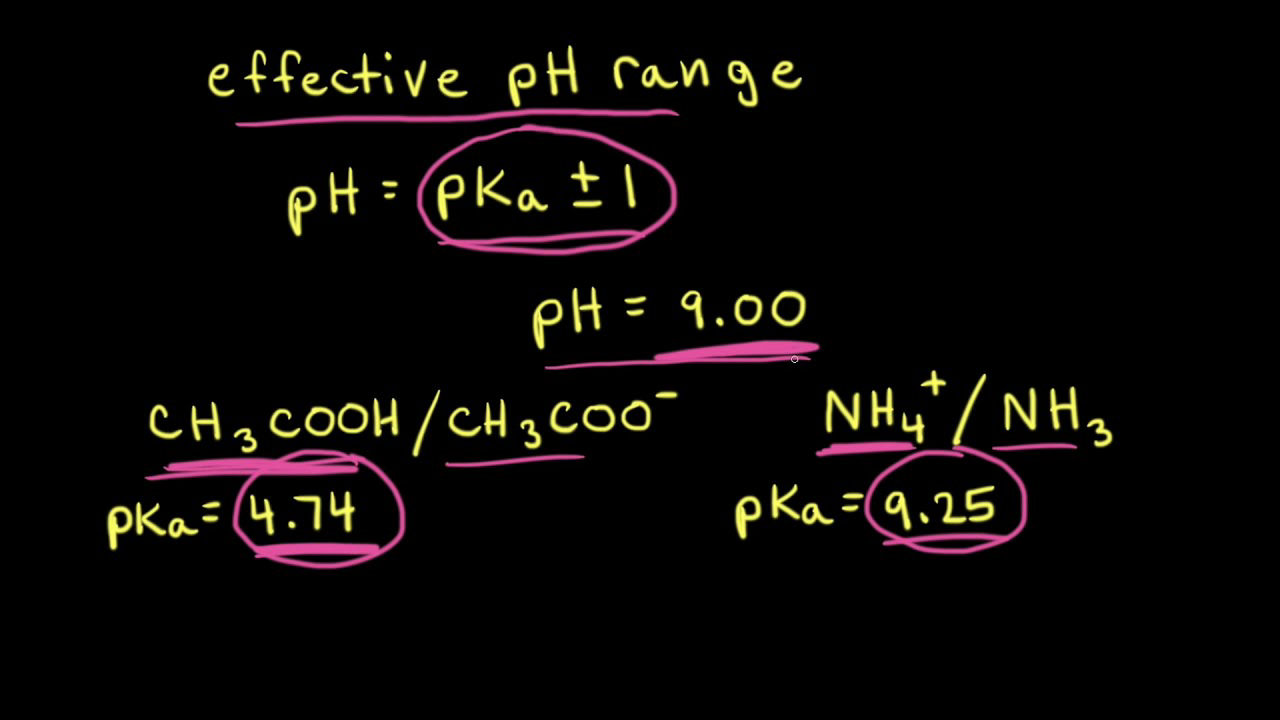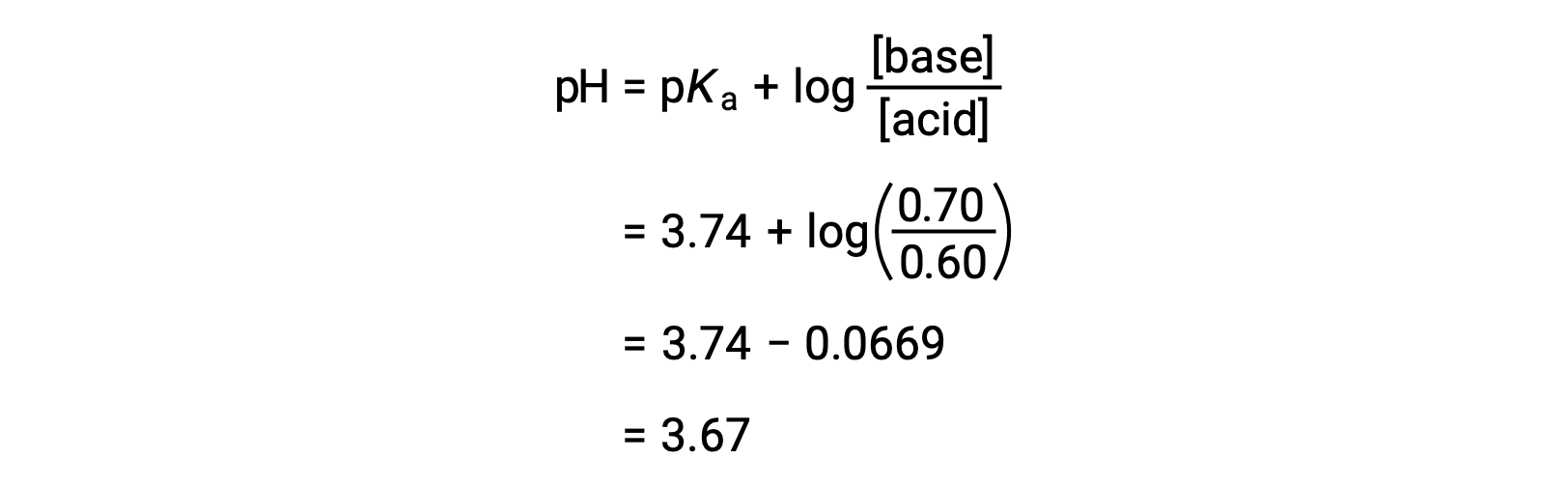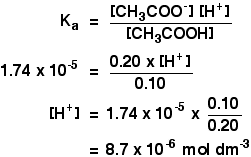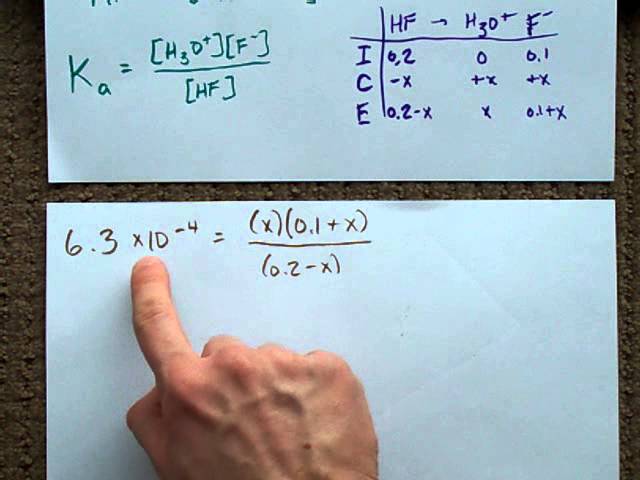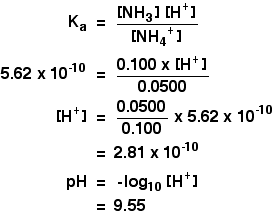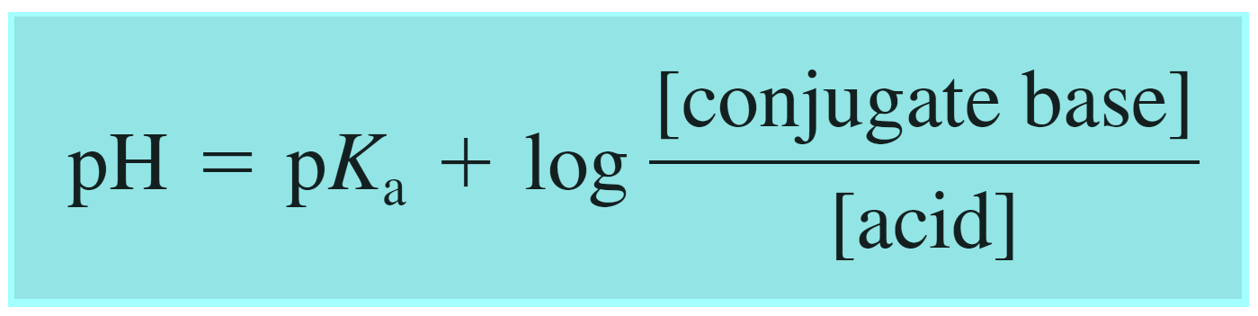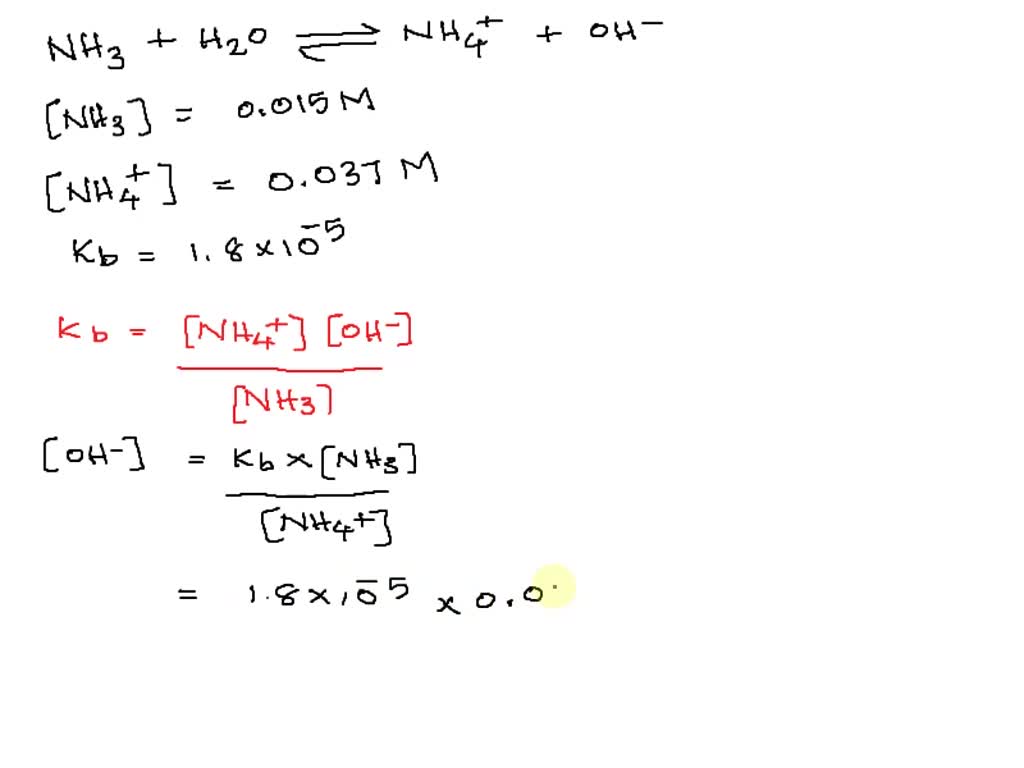
SOLVED: Calculate the pH of a buffer solution consisting of 0.051 M NH3 and 0.037 M NH4+. The Kb for NH3 = 1.8 x 10-5.

acid base - How to calculate the composition of a borate buffer with a defined pH using the Henderson-Hasselbalch equation? - Chemistry Stack Exchange

OneClass: I need help on this problem please!! B. pH of Buffer Solutionssodkum 1. Preparation of Acet...